
Download Download & share this Knowledge card in your network [Free Download]
FOLATE (Vitamin B9)
Vitamin B9, also referred to as folate or folic acid, is critical water-soluble B-vitamin that plays a vital role in various bodily functions. It’s particularly important for human beings due to its involvement in several key processes:
1. Cell Division and DNA Synthesis:
Folate is essential for the synthesis and repair of DNA, which is crucial for cell division and growth. This is especially important during periods of rapid cell division, such as during pregnancy and infancy.
2. Pregnancy and Fetal Development:
Folate is especially crucial during pregnancy. This vitamin helps prevent neural tube defects in the developing fetus. Neural tube defects are serious birth defects that affect the brain and spinal cord. Pregnant women are often advised to take folic acid (synthetic form) supplements to ensure they get enough of this vital nutrient.
3. Neurological Health:
Folate is involved in the production of neurotransmitters, which transmit signals in the brain and nervous system. Adequate folate levels are critical for proper neurological function. In adequate folate levels increase the risk of neurodegenerative disorders and neurodevelopmental disorders such as Alzheimer’s disease and basic cognitive decline.
4. Red Blood Cell Formation:
Folate is necessary for the formation and maturation of red blood cells. A deficiency in folate can lead to a type of anemia called megaloblastic anemia, characterized by large, immature red blood cells that cannot efficiently carry oxygen.
5. Heart Health:
Folate plays a role in breaking down homocysteine, an amino acid that, when elevated, is associated with an increased risk of cardiovascular disease. Adequate folate intake helps maintain lower homocysteine levels and supports heart health.
6. Mood Regulation:
Folate is involved in the synthesis of serotonin, a neurotransmitter that plays a role in regulating mood and emotions. Low levels of folate have been linked to an increased risk of depression and other mood disorders.
Clearly, Vitamin B9 is a necessary nutrient, and we must ensure that we have enough in our biological systems so as to prevent many neurological disorders. Numerous dietary sources of folate exist, including green leafy vegetables such as spinach, lettuce, broccoli, asparagus, as well as citrus fruits, beans, legumes, fortified cereals, and beef liver. In cases where dietary intake might be insufficient, supplementation or fortified foods can help ensure adequate folate intake.
Because of vitamin B9’s great importance, various items in our food supply have been preemptively fortified with folic acid, a synthetic derivative of folate (vitamin B9). Folic acid may be found in breads and cereals that are commercially available throughout the United States. The logic behind this was to fortify regular food staples so that those becoming pregnant would already have enough folate in their diet and decrease the risk of having a child with a neural tube defect. Statistically, this has helped decrease the number of neural tube defect births.
Download Download & share this Knowledge card in your network [Free Download]
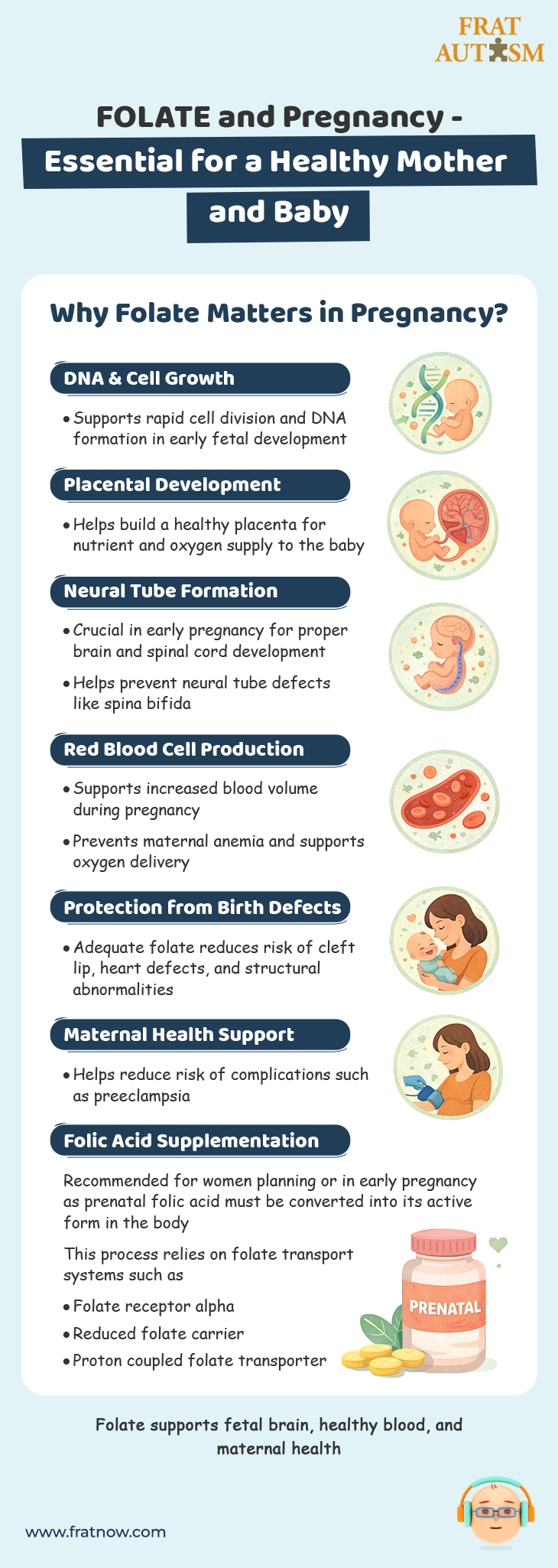
FOLATE and Pregnancy
Folate consumption during pregnancy is very important and its impact on both the mother’s health and overall fetal development is very well defined.
1. DNA Synthesis and Cell Division:
Folate is necessary for the synthesis and repair of DNA and RNA, which are essential for cell division and growth. This is particularly important during early pregnancy, when there is rapid cell division to support the development of the fetus.
2. Placental Health:
Folate is important for the formation and functioning of the placenta, which is the organ that provides necessary nutrients, oxygen, and waste removal for the developing fetus. A healthy placenta is crucial for proper fetal growth and development.
3. Neural Tube Development:
Adequate folate intake before and during early pregnancy is crucial for the proper development of the neural tube, which later forms the baby’s brain and spinal cord. Inadequate folate intake during this period can lead to neural tube defects, such as spina bifida and anencephaly. These are very serious birth defects that affect the baby’s central nervous system.
4. Other Birth Defects:
Folate deficiency has also been associated with an increased risk of other birth defects such as cleft lip and palate, heart defects, and other structural abnormalities in the baby’s organs.
5. Red Blood Cell Formation:
During pregnancy, a woman’s blood volume increases to support the needs of the developing fetus. Folate is essential for the production of red blood cells, which transport oxygen throughout the body. Inadequate folate intake can lead to anemia, which can negatively impact both the mother’s and the baby’s health.
6. Maternal Health:
Folate is also important for the mother’s health during pregnancy. It helps prevent preeclampsia, a condition characterized by high blood pressure and potential organ damage, which can be harmful to both the mother and the baby.
Due to these critical roles, healthcare providers typically recommend that women who are planning to become pregnant or are in the early stages of pregnancy take folic acid supplements. Prenatal vitamins contain Vitamin B-9, typically in its stable, oxidized form, rather the reduced form (the naturally occurring form is methylated). The synthetic version, which is found in the majority of prenatal vitamins, needs to be further reduced by the body and transported into the appropriate cells via the folate receptor alpha, the reduced folate carrier or the proton coupled folate transporter – the main receptors on cells to make reduced folate functional.
FOLATE, Folate Receptor Autoantibodies and exposure in families
Most individuals can convert sufficient folic acid to folate. However, a fairly recent discovery of autoantibodies to the folate receptor alpha (one of the main folate transporters in cells) indicates that approximately 10% of the population is limited in absorbing folate into the brain because of the presence of these folate receptor autoantibodies (FRAs). Folate Receptor Autoantibodies (FRAs) block high affinity folate receptors, rendering them dysfunctional and preventing folate from crossing the blood brain barrier. These folate receptor autoantibodies have been found in Cerebral Folate Deficiency Syndrome, Autism Spectrum Disorders, Neural Tube Defects, Depression and Schizophrenia, just to name a few neuropsychiatric/neurodevelopmental disorders.
Ultimately, those with FRAs have a reduced amount of folate reaching their brain, creating a condition known as Cerebral Folate Deficiency Syndrome (CFDS). Studies have shown that a large percentage of ASD children have these folate receptor autoantibodies, suggesting an impaired transport of folate into the brain.
While FRAs are present in a small subsection of the population, clinical tests find that about 70% of children with ASD have FRAs. Meta-analysis from multiple studies show that children with ASD are 20 times more likely to have FRAs, pointing to a likely genetic component that restricts brain absorption of folate (Vitamin B-9).
Parents of autistic children also have FRAs at substantially higher rates than the general public, which may play a critical role during neurodevelopmental critical periods in the fetus.
A 2018 study by Dr. Edward Quadros of SUNY Downstate revealed that 76% of the affected children, 75% of the unaffected siblings, 69% of fathers and 59% of mothers were positive for FRAs, whereas the prevalence of FRAs in the normal controls was 29%. FRAs were highly prevalent in affected families including unaffected siblings. The appearance of these antibodies may have a familial origin. The antibody response appears heritable with FRAs in the parents and affected child increasing the risk of ASD.
Additionally, another previous study examining the prevalence of FRAs in parents of children with ASD showed that about 26% of the mothers and 18% of the fathers were positive for FRAs compared to only 3% of parents with normal children. It is thought that maternal autoantibody titers and folate status during pregnancy and a similar disposition in the infant could influence neurodevelopmental outcome. It is likely that the pathologic effects of the FRAs are exerted early on during fetal development and during early infancy.
Besides fetal exposure to maternal folate receptor autoantibodies, paternal FRAs may exert their influence on the imprinting genes and by various epigenetic modifications. The male involvement in contributing to FRAs during conception has never been investigated. However, folate status in male conception is a well-known and a critical component. Research in this area is exceedingly necessary as we may be missing half the picture. Could it be that the male also contributes to folate receptor autoantibodies in the fetus?
The promising aspect to ascertaining the presence of folate receptor autoantibodies is that a corrective measure may be available to help with overall prognosis.
In clinical trials, children with ASD, who were positive for FRAs, showed vast improvement in their communication and language skills when placed on a daily supplement of folate in its reduced form (folinic acid). The reasoning for this is that a reduced folate, such as folinic acid, is able to enter the brain via the reduced folate carrier, which is an alternate receptor that is available. The reduced folate carrier, however, has a lower affinity for folate, therefore it needs greater folate saturation. Nonetheless, marked improvement has been seen in children that have these autoantibodies and have been administered folinic acid in larger doses (typically 1mg/per kilogram of body weight). Supplementing reduced folate earlier in development, including in utero, may be most effective in reducing the severity of ASD symptoms by facilitating typical passage of this vitamin through critical neurodevelopmental periods.
With this in mind, could it be that ASD may result from CFDS during the critical period of development? In other words, can it be because of an impediment in folate transport, whereas folate receptor autoantibodies render the folate receptor alpha dysfunctional and not allow enough folate to be transported into the brain, or the placenta during pregnancy? Could it be that the presence of FRAs in the mother blocks folate delivery to the developing fetus? Identifying pregnancies where FRAs are present may be a good practice, whereas it may also make sense to treat women who are positive with folinic acid or methylfolate.
Additionally, there may be an even greater advantage in diagnosing FRAs in prospective parents as a whole. The possibility of supplementing the parents who are positive for the FRAs even before pregnancy occurs, and the mother throughout the pregnancy with folinic acid to prevent folate deficiency in the fetus, may present a valid course of action. Therefore, identification of parents and infants with FRAs, provides the potential for early intervention to prevent or reduce the risk of ASD in the offspring.
For information on autism monitoring, screening and testing please read our blog.
References
- Bobrowski Khoury N, Ramaekers VT, Sequeira JM, Quadros EV (2021) Folate Receptor Alpha Autoantibodies in Autism Spectrum Disorders: Diagnosis, Treatment and Prevention. J Pers Med 11(8): 710.
- Frye RE, Sequeira JM, Quadros EV, James SJ, Rossignol DA (2013) Cerebral folate receptor autoantibodies in autism spectrum disorder. Mol Psychiatry 18(3): 369-381.
- Frye RE, Slattery J, Delhey L, B Furgerson, T Strickland, et al. (2018) Folinic acid improves verbal communication in children with autism and language impairment: a randomized double-blind placebo-controlled trial. Mol Psychiatry 23(2): 247-256.
- Quadros EV, Sequeira JM, Brown WT, Clifford Mevs, Elaine Marchi, et al. (2018) Folate receptor autoantibodies are prevalent in children diagnosed with autism spectrum disorder, their normal siblings and parents. Autism Research 11(5): 707-712.
- Ramaekers VT, Sequeira JM, DiDuca M, Géraldine Vrancken,1Aurore Thomas, et al. (2019) Improving Outcome in Infantile Autism with Folate Receptor Autoimmunity and Nutritional Derangements: A Self-Controlled Trial. Autism Research and Treatment 2019: 7486431.
- Ramaekers VT, Rothenberg SP, Sequeira JM, Thomas Opladen, Nenad Blau, et al. (2005) Autoantibodies to folate receptors in the cerebral folate deficiency syndrome. N Engl J Med 352(19): 1985-1991.
- Rossignol DA, Frye RE (2021) Cerebral Folate Deficiency, Folate Receptor Alpha Autoantibodies and Leucovorin (Folinic Acid) Treatment in Autism Spectrum Disorders: A Systematic Review and Meta-Analysis. J Pers Med 11(11): 1141.
- Shapira I, Sequeira JM, Quadros EV (2015) Folate receptor autoantibodies in pregnancy related complications. Birth defects research. Birth Defects Res A Clin Mol Teratol 103(12): 1028-1030.




