Table of Contents
- Introduction
- The Element That Ignites Worlds and Sustains Life
- Phosphorus — The Element That Brings Light to Life
- Cosmic Element — Forged in Stars, Delivered by Meteorites
- Element of Life — The Universal Currency of Energy and Genetics
- Food Element — Abundant, Essential, and Widely Distributed
- Dangerous Element — When Phosphorus Turns Lethal
- Medical Element — From Dubious Tonics to Modern Therapeutics
- Element of Surprises — The Unpredictable Nature of Phosphorus
- Take-Home Messages
- Summary and Conclusions
- Did You Know About Folate Receptor Autoantibodies (FRAAs) and Brain Development?
- References
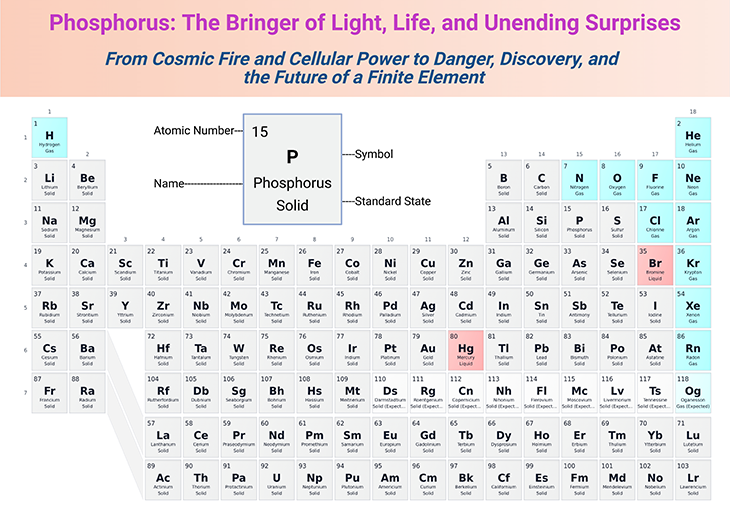
Figure 1. Phosphorus: The Bringer of Light, Life, and Unending Surprises. From cosmic fire and cellular power to danger, discovery, and the future of a finite element. [Adapted and modified from: https://pubchem.ncbi.nlm.nih.gov/periodic-table/]
Introduction
The Element That Ignites Worlds and Sustains Life
Few elements carry a story as dramatic, paradoxical, and far-reaching as phosphorus. Its very name—drawn from the Greek phosphoros, meaning “bringer of light”—once referred to the radiant Morning Star, Venus, shimmering in the dawn sky. Yet phosphorus does far more than glow; it illuminates the history of our planet, the architecture of our cells, and the chemistry that powers every heartbeat and thought. It is an element that can ignite spontaneously, poison silently, nourish entire ecosystems, and fuel the molecular engines that sustain life. In phosphorus, nature has woven together creation and destruction, stability and volatility, cosmic origins and biological necessity.
Phosphorus is a cosmic gift, forged in the fiery cores of massive stars and delivered to Earth through meteorites that have rained down for billions of years. These celestial messengers have supplied nearly 100 billion tons of phosphorus over geological time, replenishing an element that life constantly consumes and that oceans continually bury. Without this steady cosmic contribution, Earth’s biosphere would have long ago exhausted one of its most essential ingredients [1-3, 7].
Yet the true magic of phosphorus emerges when it binds with oxygen to form phosphate (PO₄³⁻)—the safe, stable, and exquisitely versatile molecule that underpins the chemistry of life. Phosphate forms the backbone of DNA, energizes cells through ATP, shapes membranes through phospholipids, and mineralizes the skeleton that supports the human body [8-10]. Every hour, the human metabolism produces, uses, and recycles more than 1 kg of ATP, a staggering testament to phosphate’s central role in biology. During intense exertion, this demand can soar to 1 kg every two minutes, revealing just how deeply life depends on this single molecular motif.
But phosphorus is not merely an element of life—it is also an element of danger, surprise, and scientific wonder. In its elemental form, it can burn through flesh or glow eerily in the dark. In industrial history, it caused the devastating disease phossy jaw. In warfare, its organophosphate derivatives became some of the most lethal nerve agents ever created. And in modern medicine, its chemistry has been transformed into life-saving therapies that strengthen bone, fight cancer, and modulate immunity.
Phosphorus is, in every sense, an element of extremes: a bringer of light, a builder of life, a harbinger of danger, and a source of endless scientific fascination. Its story spans the stars, the soil, the cell, and the human imagination—making it one of the most captivating elements in the periodic table (see Figure 1).
Phosphorus — The Element That Brings Light to Life
Pronounced fos-for-us, this element takes its name from the Greek phosphoros, meaning bringer of light—a title also bestowed upon the Morning Star, Venus. The name is fitting: phosphorus has illuminated both the night sky and the story of life on Earth. In its safest and most biologically essential form, phosphorus appears as phosphate (PO₄³⁻), a structure in which one phosphorus atom is surrounded by four oxygen atoms. In this configuration, phosphate is stable, indispensable, and foundational to global biogeochemical cycles. Yet when these oxygen atoms are stripped away, phosphorus becomes reactive and dangerous, unleashing a chemistry that is far more volatile.
Cosmic Element — Forged in Stars, Delivered by Meteorites
Phosphorus is a true cosmic element, born in the hearts of massive stars—far larger than our Sun. Its nucleus forms when two oxygen nuclei fuse, followed by the loss of a proton, yielding the atomic number 15 (8 + 8 – 1 = 15). Much of the phosphorus present on Earth today arrived via meteorites, which continue to deliver roughly 100 tons of extraterrestrial material to our planet each day. With an average phosphorus content of 0.1%, primarily as the mineral schreibersite, this influx contributes about 35 tons of phosphorus per year. Over Earth’s 4.6-billion-year history, meteorites have supplied an estimated 100 billion tons of phosphorus—an essential replenishment for an element continually lost to the deep ocean through sedimentation [1-3, 7].

Table 1. Element of Life ~ Phosphorus in the Human Body.
Element of Life — The Universal Currency of Energy and Genetics
In nature, phosphorus is never found as the pure element; it exists exclusively as phosphate. In minerals, phosphate appears as the negatively charged PO₄³⁻ ion, while in living organisms it forms organophosphates, where organic molecules attach to one or more oxygen atoms. In human blood, 92% of phosphorus circulates as organophosphate, with only 8% present as free phosphate. Adult blood typically contains about 30 ppm of phosphate, whereas children have nearly double, reflecting the heightened demand for growth and skeletal development [8-10] (see Table 1).
Although the brain contains the highest concentration of phosphate relative to its mass, the skeleton holds the largest total amount, as bone is composed largely of calcium phosphate. Phosphate is indispensable to life: it forms the backbone of DNA, powers cellular processes through ATP (adenosine triphosphate), transmits signals via GMP (guanosine metaphosphate), and builds the phospholipid membranes that encase every cell. The scale of ATP turnover is astonishing—more than 1 kg of ATP is produced, used, and recycled every hour, fueled by glucose metabolism. During intense physical exertion, ATP demand can surge to 1 kg every two minutes, underscoring the centrality of phosphate to human physiology [4].
Because DNA, ATP, GMP, and most organophosphates carry negative charges, they must be balanced by positively charged ions. The preferred partner is magnesium (Mg²⁺), which stabilizes these molecules and enables their biological function.
Food Element — Abundant, Essential, and Widely Distributed
Phosphate is a dietary necessity, with a recommended intake of 800 mg per day. Fortunately, most individuals easily exceed this requirement: typical diets supply 1,000–2,000 mg per day, depending on the consumption of phosphate-rich foods. Many animal-derived foods contain more than 200 mg per 100 g, including tuna, salmon, sardines, liver, turkey, chicken, eggs, and cheese.
Natural phosphorus content varies widely. Liver can contain up to 370 mg per 100 g, while vegetable oils contain almost none. Lean meats average 180 mg per 100 g, and eggs provide about 220 mg per 100 g. Some processed cheeses contain as much as 500 mg per 100 g, largely due to the addition of 2% disodium phosphate, which prevents fat separation. Other processed foods—such as sausages and cooked meats—also incorporate phosphate additives.
However, not all phosphate-rich foods provide digestible phosphorus. Plants store phosphate as inositol hexaphosphate (IHP), a compound that supports seed germination but is poorly digestible in humans. For example, wholemeal flour contains 340 mg phosphate per 100 g, but much of this is IHP. In contrast, white flour contains 130 mg per 100 g, most of which is digestible.
Phosphate additives, considered safe, are widely used in the food industry. Common additives include sodium, calcium, and potassium salts of phosphoric acid (H₃PO₄)—which itself is added to cola beverages. Disodium phosphate stabilizes evaporated milk, preventing it from becoming semi-solid during storage. Phosphate additives first appeared in the 19th century as leavening agents, combining calcium dihydrogen phosphate with sodium bicarbonate to lighten cakes and pastries. These compounds remain the key ingredients in self-raising flour.
Approved phosphate additives carry E-codes:
- E339 — sodium phosphates
- E340 — potassium phosphates
- E341 — calcium phosphates
- E343 — magnesium phosphates
- E342 — ammonium phosphates (restricted in some countries)
- E338 — phosphoric acid
The human skeleton serves as a vast phosphate reservoir, continuously remodeled throughout life. Bone is far from inert; it is constantly deposited and resorbed at millions of microscopic sites. When dietary intake is insufficient, the body draws on this reserve. A deficiency of phosphate—known as hypophosphatemia—can arise from inadequate intake, impaired absorption, or excessive losses, and can disrupt energy metabolism, bone integrity, and cellular function.
Dangerous Element — When Phosphorus Turns Lethal
Among the many faces of phosphorus, none is more infamous than white phosphorus, the elemental form most widely recognized. Highly reactive, dangerously flammable, and profoundly toxic, white phosphorus can be lethal in quantities as small as 100 mg. Once ingested, it rapidly targets the liver, triggering catastrophic organ failure that typically leads to death within one week. Its volatility extends to inhalation: prolonged exposure to phosphorus vapor once caused the devastating industrial disease known as phossy jaw, a condition that slowly destroyed the jawbone. This affliction haunted workers in the 19th-century match industry, and sporadic cases continued into the 20th century among those handling the substance.
Transporting white phosphorus poses its own hazards. Because it must be kept molten, it is shipped as a liquid, and accidents have been severe. On 1 April 1978, a rail tanker carrying 50,000 liters of molten phosphorus ruptured in Brownston, Nebraska, igniting a fire that burned for three days and destroyed 30 acres of crops. A similar derailment occurred near Lviv, on the Ukraine–Poland border, when a freight train transporting phosphorus caught fire—fortunately without casualties. In contrast to white phosphorus, red phosphorus is far safer: it is non-flammable and produced by heating white phosphorus in a sealed vessel for several days, transforming its structure into a more stable polymeric form.
Beyond elemental phosphorus, certain organophosphates pose extreme danger. These compounds can irreversibly inhibit acetylcholinesterase, the enzyme responsible for terminating nerve impulses at synapses. When this enzyme is blocked, acetylcholine accumulates, causing uncontrolled stimulation of muscles and organs, ultimately leading to respiratory failure and death. This mechanism underlies the lethality of organophosphate chemical warfare agents, including the notorious tabun and sarin.
Medical Element — From Dubious Tonics to Modern Therapeutics
Despite its dangers, phosphorus once enjoyed a prominent—if misguided—place in medicine. In the 19th century, texts such as Free Phosphorus in Medicine (1874) praised its supposed therapeutic virtues. Physicians prescribed minute doses—typically 1/20th of a grain (≈3 mg)—for an astonishing range of conditions: nervous breakdown, depression, migraine, epilepsy, psychiatric disorders, and even erectile dysfunction. It was also recommended for physical ailments including stroke, pneumonia, alcoholism, tuberculosis, cholera, and cataracts. Phosphorus-based tonics and toothache remedies were sold over the counter well into the early 20th century, and medical prescriptions persisted into the 1920s, though by 1930 elemental phosphorus had been removed from official pharmacopoeias.
A supposedly safer alternative emerged in the form of hypophosphorous acid (H₃PO₂) and its salts, the hypophosphites, which gained popularity after 1857. These compounds were marketed for conditions such as neurasthenia—a diagnosis characterized by fatigue and low vitality—and even for tuberculosis, though they offered no real therapeutic benefit. One of the most enduring formulations was Scott’s Emulsion, a mixture of cod liver oil and calcium hypophosphite, sold for decades as a general restorative.
Modern medicine, however, has found legitimate therapeutic uses for phosphorus-containing compounds. Bone health depends on the dynamic interplay between osteoblasts, which build bone, and osteoclasts, which break it down. With age, bone resorption often outpaces formation, leading to osteoporosis and increased fracture risk. To counter this, clinicians use bisphosphonates, synthetic molecules composed of two phosphate groups linked through a carbon atom—[O₃P–C–PO₃]⁴⁻—with two additional substituents on the carbon. These drugs bind strongly to bone mineral, inhibiting osteoclast activity and slowing bone loss. Bisphosphonates are also used to treat Paget’s disease and bone metastases. While some patients report side effects such as joint pain, emerging research suggests that certain bisphosphonates may also enhance immune function and induce cancer cell death, expanding their therapeutic relevance [5].
Another biologically important class of phosphorus compounds is the polyphosphates—long chains of phosphate units linked together. These inorganic polymers play a crucial role in blood clotting, as platelets release polyphosphates at sites of injury to accelerate coagulation and stabilize forming clots.
Element of Surprises — The Unpredictable Nature of Phosphorus
Phosphorus has long captivated the human imagination, not only for its essential role in biology but also for its uncanny ability to behave in ways that seem almost supernatural. In Arthur Conan Doyle’s The Hound of the Baskervilles, the terrifying spectral dog owes its ghostly glow to a coating of white phosphorus, a theatrical trick that Victorian audiences found both mesmerizing and macabre. Historical accounts suggest that some individuals in the Victorian era even applied dilute white phosphorus solutions to their skin to create a similar eerie luminescence—an early, dangerous form of special effects.
Beyond its dramatic flair, phosphorus continues to surprise in far more consequential ways. Around the world, environmental regulations now require the removal of phosphate from sewage, a shift driven by concerns over eutrophication and water quality. This mandate has opened the door to a remarkable possibility: that nearly all the phosphate needed for agriculture and industry could one day be reclaimed from wastewater. Modern treatment technologies can extract phosphate efficiently, and in many respects this reclaimed material is cleaner than phosphate rock, which often contains contaminants such as cadmium and uranium that must be removed before use. In the future, the phosphates used in dishwasher detergents, processed foods, and even fertilizers may originate from the waste that flows through our kitchen sinks and bathroom drains—a striking example of circular chemistry.
Phosphorus also defies expectations at the level of fundamental physics. In 2004, Japanese researchers discovered that molten red phosphorus, when heated to 1,000°C under pressure, could exist as two distinct liquids simultaneously. This phenomenon—liquid–liquid phase separation—was once thought impossible because liquids, like gases, are typically homogeneous, with molecules constantly intermingling. Yet in this extraordinary state, droplets of high-density liquid phosphorus floated within a lower-density liquid, revealing a complexity in phosphorus chemistry that scientists had never anticipated [6].
The element’s surprises extend into the geological past as well. Beginning in 1862, hundreds of workers in Bedfordshire and neighboring English counties made their living by excavating fossilized dinosaur bones for use in fertilizer production. These fossils lay about one meter below the soil surface in a two-meter-deep bed of rock stretching more than 100 miles along what had once been a coastal plain. The bones belonged to creatures that perished in a catastrophic event—likely linked to a meteor impact—around 95 million years ago. Their phosphorus-rich remains became a valuable agricultural resource, illustrating how the element cycles through life, death, and deep time.
Even at the molecular level, phosphorus continues to astonish. In 2010, chemists succeeded in producing the rare and highly reactive molecule P₂, a diatomic form of phosphorus long predicted but never previously isolated. Its fleeting existence offers new insights into phosphorus bonding and reactivity, expanding our understanding of an element that refuses to conform to expectations.
Take-Home Messages
- Phosphorus is a cosmic element, forged in massive stars and delivered to Earth through meteorites that have supplied nearly 100 billion tons over geological time.
- Life depends on phosphate (PO₄³⁻)—the safe, oxygen-bound form of phosphorus that builds DNA, fuels cells through ATP, stabilizes membranes, and forms the mineral foundation of bone.
- Human metabolism recycles more than 1 kg of ATP every hour, and during intense exertion this demand can rise to 1 kg every two minutes, underscoring phosphate’s central role in energy flow.
- Dietary phosphate is abundant, with typical intake far exceeding the recommended 800 mg/day, though plant-derived inositol hexaphosphate (IHP) remains poorly digestible.
- White phosphorus is dangerously reactive and highly toxic, historically causing industrial diseases like phossy jaw and posing major risks during transport and handling.
- Organophosphate nerve agents, such as tabun and sarin, exploit phosphorus chemistry to block acetylcholinesterase, leading to fatal disruption of nerve signaling.
- Modern medicine harnesses phosphorus safely through compounds like bisphosphonates, which bind bone mineral, inhibit osteoclasts, and treat osteoporosis, Paget’s disease, and bone cancers.
- Environmental recovery of phosphate from sewage is emerging as a sustainable, contaminant-free source that may one day replace mined phosphate for agriculture and industry.
- Phosphorus continues to surprise science, from the discovery of dual-liquid red phosphorus at 1,000°C to the isolation of the rare diatomic molecule P₂ in 2010.
(Cf. previous blogs entitled as: “Iodine: The Violet Element That Shapes the Human Mind.”; “Selenium: The Moon-Named Element That Protects Human Life.”; “Zinc: The Essential Element We Cannot Live Without.”)
Summary and Conclusions
Phosphorus stands as one of the most paradoxical and indispensable elements in the natural world—an element forged in the hearts of massive stars, delivered to Earth by meteorites, and woven into the very architecture of life. Its stable form, phosphate (PO₄³⁻), is the molecular backbone of DNA, the energy currency of ATP, the structural foundation of bone, and the membrane-forming scaffold that defines every living cell. Human metabolism depends on phosphate with astonishing intensity, recycling more than 1 kg of ATP per hour, and far more during strenuous exertion. Yet this same element, when stripped of oxygen, becomes dangerously reactive: white phosphorus can ignite spontaneously, poison the liver in milligram doses, and historically caused the devastating industrial disease phossy jaw. This duality—life-giving and lethal—makes phosphorus one of the most compelling elements in the periodic table.
Despite its abundance in the diet and its centrality to biology, phosphorus presents ongoing challenges for human health, agriculture, and the environment. Modern societies rely heavily on mined phosphate rock, yet these deposits often contain contaminants such as cadmium and uranium, and global reserves are finite. This has driven a new era of innovation in phosphate recovery from sewage, where advanced technologies now reclaim phosphorus in forms cleaner than those extracted from rock. Such circular systems may one day supply nearly all the phosphate needed for farming, industry, and food production. At the same time, the medical field has transformed phosphorus chemistry into powerful therapeutics: bisphosphonates strengthen bone, treat Paget’s disease, and slow bone metastases, while emerging research suggests they may also modulate immunity and induce cancer cell death. Yet gaps remain in understanding their long-term effects, optimal dosing strategies, and the mechanisms underlying rare but serious side effects.
Phosphorus continues to astonish scientists with behaviors once thought impossible. The discovery in 2004 that molten red phosphorus can exist as two distinct liquids at 1,000°C challenged fundamental assumptions about the nature of matter. The successful isolation of the elusive P₂ molecule in 2010 opened new windows into phosphorus bonding and reactivity. Even the geological record reveals phosphorus’s surprising legacy: vast beds of fossilized dinosaur bones—formed after a catastrophic event 95 million years ago—once served as fertilizer sources across England, illustrating how the element cycles through deep time, ecosystems, and human industry.
Looking ahead, several knowledge gaps and future directions emerge. We still lack a complete understanding of how phosphorus cycles through soils under climate-driven changes, how microplastics and emerging pollutants influence phosphate availability, and how global agriculture can transition from finite mined sources to sustainable recovery systems. In medicine, deeper insights are needed into how phosphorus-containing drugs interact with bone remodeling pathways, immune signaling, and cancer biology. And at the molecular level, the unusual behaviors of phosphorus allotropes and reactive intermediates continue to challenge chemists, suggesting that this element still holds secrets waiting to be uncovered.
Ultimately, phosphorus is far more than a nutrient or industrial commodity—it is a cosmic inheritance, a biological necessity, a chemical enigma, and a driver of both innovation and risk. Its story spans the stars, the soil, the cell, and the human imagination. As we continue to explore its mysteries and harness its potential, phosphorus reminds us that even the most familiar elements can surprise us, illuminate new scientific frontiers, and shape the future of life on Earth.
For information on autism monitoring, screening and testing please read our blog.
References
- Elser JJ, Bracken ME, Cleland EE, Gruner DS, Harpole WS, Hillebrand H, Ngai JT, Seabloom EW, Shurin JB, Smith JE. Global analysis of nitrogen and phosphorus limitation of primary producers in freshwater, marine and terrestrial ecosystems. Ecol Lett. 2007 Dec;10(12):1135-42. doi: 10.1111/j.1461-0248.2007.01113.x. Epub 2007 Oct 6. PMID: 17922835.
https://pubmed.ncbi.nlm.nih.gov/17922835/
(A foundational synthesis on phosphorus as a limiting nutrient across Earth’s ecosystesm.) - Cordell D, Drangert JO, White S. The Story of Phosphorus: Global Food Security and Food for Thought. May 2009 Global Environmental Change 19(2):292-305. DOI:10.1016/j.gloenvcha.2008.10.009.
https://www.sciencedirect.com/science/article/abs/pii/S095937800800099X
https://www.diva-portal.org/smash/get/diva2:291760/FULLTEXT01.pdf
(A landmark paper on phosphorus scarcity, sustainability, and future recovery strategies.) - Filippelli, Gabriel. (2008). The Global Phosphorus Cycle: Past, Present, and Future. Elements. 4. 89-95. 10.2113/GSELEMENTS.4.2.89.
https://pubs.geoscienceworld.org/msa/elements/article-abstract/4/2/89/137768/The-Global-Phosphorus-Cycle-Past-Present-and?redirectedFrom=fulltext
(A concise, authoritative overview of phosphorus cycling from geological to human timescales.) - Bird RP, Eskin NAM. The emerging role of phosphorus in human health. Adv Food Nutr Res. 2021;96:27-88. doi: 10.1016/bs.afnr.2021.02.001. Epub 2021 Apr 15. PMID: 34112356.
https://pubmed.ncbi.nlm.nih.gov/34112356/
(A clear, clinically relevant review of phosphorus physiology, metabolism, and dietary considerations.) - Russell RG, Xia Z, Dunford JE, Oppermann U, Kwaasi A, Hulley PA, Kavanagh KL, Triffitt JT, Lundy MW, Phipps RJ, Barnett BL, Coxon FP, Rogers MJ, Watts NB, Ebetino FH. Bisphosphonates: an update on mechanisms of action and how these relate to clinical efficacy. Ann N Y Acad Sci. 2007 Nov;1117:209-57. doi: 10.1196/annals.1402.089. PMID: 18056045.
https://pubmed.ncbi.nlm.nih.gov/18056045/
(A definitive paper on how phosphorus-containing bisphosphonates treat osteoporosis and bone disease.) - Katayama Y, Mizutani T, Utsumi W, Shimomura O, Yamakata M, Funakoshi K. A first-order liquid-liquid phase transition in phosphorus. Nature. 2000 Jan 13;403(6766):170-3. doi: 10.1038/35003143. PMID: 10646596.
https://pubmed.ncbi.nlm.nih.gov/10646596/
(The groundbreaking study revealing the dual-liquid behavior of molten red phosphorus at 1,000°C.) - Pasek MA, Lauretta DS. Aqueous corrosion of phosphide minerals from iron meteorites: a highly reactive source of prebiotic phosphorus on the surface of the early Earth. Astrobiology. 2005 Aug;5(4):515-35. doi: 10.1089/ast.2005.5.515. PMID: 16078869.
https://pubmed.ncbi.nlm.nih.gov/16078869/
(A pivotal paper linking meteorites, schreibersite, and the origins of bioavailable phosphorus on early Earth.) - Koljonen L, Enlund-Cerullo M, Hauta-Alus H, Holmlund-Suila E, Valkama S, Rosendahl J, Andersson S, Pekkinen M, Mäkitie O. Phosphate Concentrations and Modifying Factors in Healthy Children From 12 to 24 Months of Age. J Clin Endocrinol Metab. 2021 Sep 27;106(10):2865-2875. doi: 10.1210/clinem/dgab495. PMID: 34214153; PMCID: PMC8475199.
https://pubmed.ncbi.nlm.nih.gov/34214153/
(Shows how phosphate levels shift during rapid brain growth and identifies biological modifiers relevant to neurodevelopment.) - Cohen Kadosh K, Muhardi L, Parikh P, Basso M, Jan Mohamed HJ, Prawitasari T, Samuel F, Ma G, Geurts JM. Nutritional Support of Neurodevelopment and Cognitive Function in Infants and Young Children-An Update and Novel Insights. Nutrients. 2021 Jan 10;13(1):199. doi: 10.3390/nu13010199. PMID: 33435231; PMCID: PMC7828103.
https://pubmed.ncbi.nlm.nih.gov/33435231/
(Highlights the essential role of nutrients (including phosphorus‑dependent pathways) in early brain development.) - Valentine CJ. Nutrition and the developing brain. Pediatr Res. 2020 Jan;87(2):190-191. doi: 10.1038/s41390-019-0650-y. Epub 2019 Oct 31. PMID: 31673115.
https://pubmed.ncbi.nlm.nih.gov/31673115/
(Discusses how nutrients involved in ATP production, membrane formation, and neurotransmission shape early neurodevelopment.)