Table of Contents
- Introduction
- The Violet Element That Shapes Human Destiny
- Iodine — The Violet Element That Shapes Human Development
- Essential Element — A Micronutrient With Outsized Consequences
- Element of Life — The Thyroid’s Signature Mineral
- Dangerous Element — A Narrow Window Between Benefit and Harm
- Food Element — How Diet Determines Thyroid Health
- Global Deficiency — A Persistent Public Health Challenge
- Medical Element — Iodine’s Journey Through Medicine and Human Health
- Element of Surprises — Chemistry, Detection, and Biological Mysteries
- Take-Home Messages
- Summary and Conclusions
- Did You Know About Folate Receptor Autoantibodies (FRAAs) and Brain Development?
- References
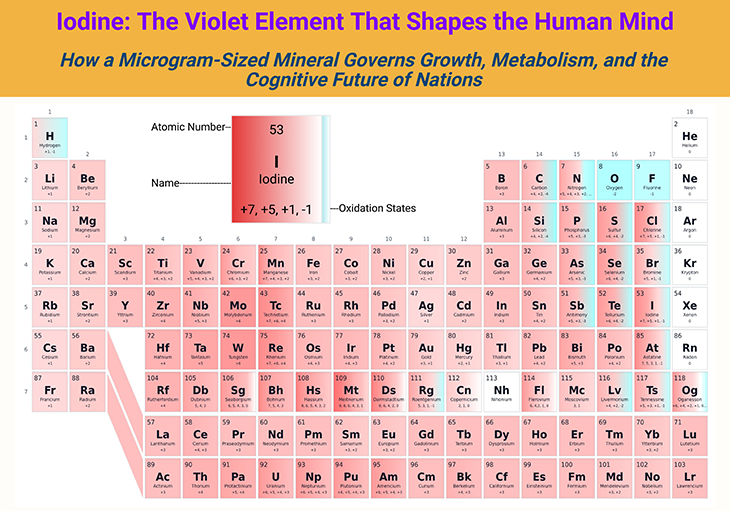
Figure 1. Iodine: The Violet Element That Shapes the Human Mind. How a microgram-sized mineral governs growth, metabolism, and the cognitive future of nations. [Adapted and modified from: https://pubchem.ncbi.nlm.nih.gov/periodic-table/]
Introduction
The Violet Element That Shapes Human Destiny
Among the elements that sustain human life, few carry a story as dramatic—or as quietly consequential—as iodine. Named from the Greek iodes, meaning “violet”, this element first revealed itself through the shimmering purple vapor rising from dark crystals. Yet behind this poetic hue lies a mineral whose influence reaches far deeper than its appearance suggests. Iodine is required in microgram quantities, but its absence has altered the intellectual landscape of entire populations, shaped public-health policy across continents, and determined the cognitive potential of generations.
For humans and animals alike, iodine is the indispensable raw material for the thyroid hormones T₄ (3,5,3’,5’-tetraiodothyronine) and T₃ (3,3’,5-triiodothyronine)—molecules that orchestrate growth, brain development, metabolic rate, and thermal balance. A deficiency during pregnancy can irreversibly impair the developing brain, while excess intake can destabilize the delicate machinery of thyroid regulation. This narrow physiological window has made iodine both a lifesaving nutrient and a potential disruptor, depending on how—and how much—is consumed.
The global struggle with iodine deficiency has been one of the most far-reaching nutritional challenges in human history. Regions with iodine-poor soil and water once saw widespread goiter, hypothyroidism, and cretinism, conditions that silently eroded health and human potential. The introduction of iodized salt in the 20th century stands as one of the most successful public-health interventions ever implemented, dramatically reducing the burden of iodine deficiency disorders across much of the world. Yet even today, millions remain at risk, reminding us that this element’s story is still unfolding [1-6] (see Figure 1).
Beyond nutrition, iodine has carved a remarkable path through medicine. From early tinctures and antiseptics to modern radioisotopes used in imaging and cancer therapy, iodine has repeatedly reinvented its role in clinical practice. Its chemistry continues to surprise—from its vivid blue reaction with starch to its ability to exist as both I⁻ and I⁺, a versatility that underpins the synthesis of thyroid hormones themselves.
In this chapter, we explore iodine as an essential element, a dangerous element, a food element, a medical element, and ultimately an element of surprises—a mineral whose influence spans biology, chemistry, medicine, and global health. Through this journey, the quiet power of iodine becomes unmistakable: a trace element with the capacity to shape the destiny of individuals, communities, and entire nations.
Iodine — The Violet Element That Shapes Human Development
Pronounced iyo-deen, this element takes its name from the Greek word iodes, meaning violet, a nod to the striking purple vapor released by crystalline iodine. In chemistry, iodine (I₂) refers to the elemental form—two iodine atoms bound together—responsible for the familiar black crystals, violet fumes, and the yellow antiseptic solutions used in medicine. When iodine gains an electron, it becomes the more stable iodide ion (I⁻). Although these forms differ chemically, the general term “iodine” is used broadly in nutrition, medicine, and environmental science whenever the specific molecular form is not central to the discussion.
Essential Element — A Micronutrient With Outsized Consequences
Despite requiring less than 0.1 mg of iodine per day, humanity has struggled with deficiency for centuries. The consequences have been devastating: millions of children have been condemned to cretinism, a condition marked by severely impaired cognitive development and extremely low IQ, all because their mothers lacked this tiny but vital element during pregnancy. Iodine deficiency remains one of the leading preventable causes of intellectual disability worldwide.
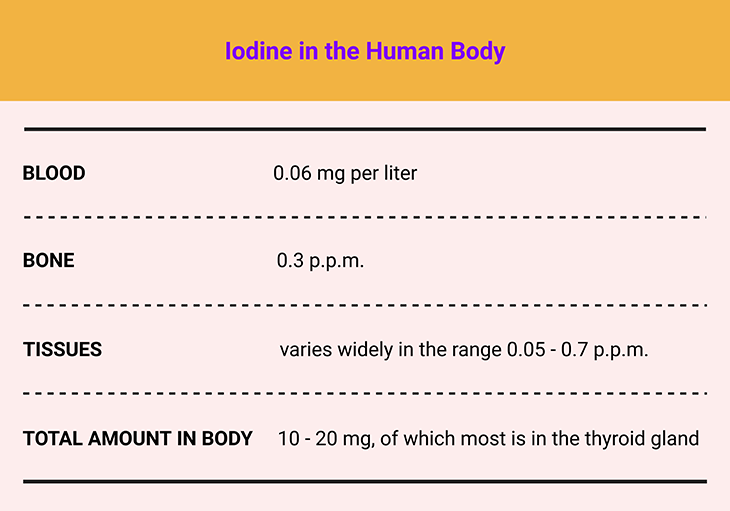
Table 1. Element of Life ~ Iodine in the Human Body.
Element of Life — The Thyroid’s Signature Mineral
Iodine is indispensable for humans and animals. Nearly all of the body’s iodine is concentrated in the thyroid gland, where it forms two essential hormones:
- 3,5,3’,5’-tetraiodothyronine (T₄ or thyroxine)
- 3,3’,5-triiodothyronine (T₃)
These hormones regulate growth, brain development, metabolic rate, and body temperature across the lifespan. Beyond the thyroid, iodine is also found in the salivary glands, stomach, pituitary, ovaries, epidermis, and components of the immune system.
Although the body can recycle iodine, small amounts are lost daily through urine, making dietary intake essential. Interestingly, iodine is not required by plants, though they readily absorb it from soil or air, which is why plant iodine content varies dramatically by geography [1-6] (see Table 1).
Dangerous Element — A Narrow Window Between Benefit and Harm
Iodine’s reactivity makes it both useful and hazardous. Iodine vapor irritates the eyes and lungs, and workplace exposure must remain below 1 mg/m³. Elemental iodine is toxic when ingested, with doses as low as 3 g capable of causing death. In contrast, iodides (I⁻) are generally safer, though excessive intake can still provoke adverse reactions, particularly in individuals with underlying thyroid disease.
Food Element — How Diet Determines Thyroid Health
Humans require a minimum of 70 μg of iodide per day, while pregnant and breastfeeding women need roughly double to support fetal and infant brain development.
Reference values include:
- UK dietary reference value: 140 μg/day
- US Institute of Medicine recommendation: 150 μg/day
- Typical US intake (2000): >200 μg/day for both men and women
- Typical Japanese intake: >300 μg/day due to high seafood and seaweed consumption
- Safe upper limit: ~1 mg/day (≈1000 μg), above which thyroid dysfunction becomes likely
Iodine deficiency is best assessed through urinary iodine concentration, with levels indicating iodine deficiency disorder (IDD).
In developed nations, dietary sources generally meet requirements. Milk is a major contributor, partly because iodine is added to cattle feed. Among plants, cabbages, onions, and mushrooms can accumulate up to 10 ppm (dry weight). The richest natural sources include cod, oysters, shrimp, herring, lobster, sunflower seeds, seaweed, and mushrooms. A 150 g portion of haddock contains approximately 300 μg of iodine [1-6].
Certain foods—such as cassava, maize, bamboo shoots, and sweet potatoes—contain goitrogens that interfere with iodine uptake. These pose a threat primarily in regions where iodine intake is already marginal.
Global Deficiency — A Persistent Public Health Challenge
In 2000, 750 million people in the developing world were affected by IDD, with 10 million suffering from cretinism. The highest-risk populations were in India and China, where centuries of intensive agriculture depleted soil iodine.
The World Health Organization (WHO) and UNICEF launched a global campaign to iodize all edible salt by 2000. Most affected countries now legally require iodization. Salt is typically fortified at 15 ppm, ensuring that a daily intake of 5 g of salt provides roughly 70 μg of iodide. Fortification uses either potassium iodide (KI) or potassium iodate (KIO₃), the latter being more stable in hot, humid climates [1-6].
Remarkably, supplying the entire world’s iodine needs would require only 175 tons per year. Yet deficiency persists. A 2007 report in The Lancet noted that 500 million people in India still suffered from IDD, with 2 million cases of cretinism—evidence that iodine deficiency remains a major global health issue.
Medical Element — Iodine’s Journey Through Medicine and Human Health
The medical story of iodine stretches back nearly 180 years, beginning long before its biochemical role was understood. Early physicians recognized that iodine could influence diseases of the thyroid, but the path from observation to scientific clarity was winding. Deficiency of this element leads to two of the most recognizable thyroid disorders: goiter, marked by a visibly enlarged thyroid gland and swollen neck, and hypothyroidism, a condition in which individuals become listless, fatigued, and unusually cold-sensitive. Conversely, excessive iodine intake—or abnormal thyroid handling of iodine—can trigger hyperthyroidism, characterized by restlessness, weight loss, and hyperactivity [1-6].
The first major medical use of iodine came in 1820, when Dr. Jean-François Coindet (1774–1834) introduced a preparation of iodine and potassium iodide dissolved in alcohol, known as tincture of iodine, as a treatment for goiter. Coindet had astutely noted that traditional remedies involved consuming seaweed ash, a naturally iodine-rich substance, and reasoned that iodine itself must be the active agent. His insight was correct, but tincture of iodine proved too harsh: patients experienced severe stomach pain due to its irritant effects, and the treatment fell out of favor.
A breakthrough came in 1829, when Jean Lugol (1786–1851) discovered that iodine dissolves readily in aqueous potassium iodide, creating what became known as Lugol’s iodine. This formulation was far less painful when applied to open wounds and became widely used as an antiseptic. Around the same time, physicians observed that regions plagued by goiter and cretinism, such as the Alps, had very low iodide levels in their water supply. Attempts to treat goiter with Lugol’s iodine were eventually abandoned because excessive iodide caused adverse effects—an early recognition of iodine’s narrow therapeutic window.
The true connection between iodine and the thyroid gland emerged in 1895, when Dr. Eugen Baumann reportedly spilled concentrated nitric acid on thyroid tissue and noticed violet fumes, revealing the presence of iodine. Yet it was not until 1916 that David Marine (1888–1976) of Ohio demonstrated conclusively that iodide supplementation could prevent and treat goiter, especially when delivered through the diet. His work laid the foundation for iodized salt, introduced in the United States in 1930, which led to the near-elimination of goiter within two decades.
Although iodine solutions failed as internal treatments for thyroid disease, they became indispensable as antiseptics. Tincture of iodine was widely used to disinfect wounds, long before its germ-killing properties were understood. Over time, gentler antiseptics replaced it, but iodine still plays a role in agriculture—for example, disinfecting cow udders after milking. Historically, when iodine was applied before milking, some entered the milk supply, inadvertently boosting dietary iodine intake.
Modern medicine has expanded iodine’s role far beyond antiseptics. Several radioactive isotopes are now essential diagnostic and therapeutic tools:
- Iodine-123 (half-life 13.2 hours)
- Iodine-125 (half-life 59.4 days)
- Iodine-131 (half-life 8.0 days)
Iodine-125, a gamma emitter, is used as a radioactive tracer and in prostate cancer therapy, where it is implanted as tiny titanium pellets. Because iodide naturally accumulates in the thyroid, radioactive isotopes can selectively destroy thyroid cancer cells while sparing other tissues.
Iodine-131, the first radionuclide used therapeutically after World War II, emits both beta and gamma rays as it decays to xenon gas. It remains a cornerstone in diagnosing and treating thyroid disorders, including hyperthyroidism and thyroid cancer.
Iodine’s diagnostic utility extends beyond the thyroid. As a heavy element, iodine effectively blocks X-rays, making iodinated compounds ideal contrast agents for imaging blood vessels, organs, and tumors.
In gynecology, Lugol’s iodine plays a critical role in colposcopy, where it stains healthy, glycogen-rich cervical tissue brown, leaving abnormal or cancerous tissue unstained, allowing early detection of cervical lesions.
Although iodine tincture has largely disappeared from household medicine cabinets, innovation continues. A modern therapy called Iodozyme is used for chronic leg ulcers. It employs a two-layer dressing that generates iodine in situ, through the oxidation of iodide by hydrogen peroxide—an elegant example of controlled, localized iodine release.
Element of Surprises — Chemistry, Detection, and Biological Mysteries
Iodine’s chemistry has long fascinated scientists. Its dramatic blue reaction with starch once made it indispensable in analytical titrations. This reaction still has practical use today: banknote detection pens contain Lugol’s solution, which turns blue when it contacts starch in counterfeit paper. Genuine banknote paper contains no starch, so no color change occurs.
Beyond forming the familiar iodide ion (I⁻), iodine can also lose an electron to form the positive ion (I⁺). This less common oxidation state may play a role in the biosynthesis of organo-iodine compounds, including the formation of thyroxine (T₄) within the thyroid gland—an area where biochemical research continues to evolve.
Take-Home Messages
- Iodine is essential for human biology, yet required in only microgram amounts each day.
- The thyroid concentrates nearly all the body’s iodine, using it to produce the hormones T₄ and T₃ that regulate growth, metabolism, and temperature.
- Even mild deficiency during pregnancy impairs fetal brain development, making iodine one of the most critical nutrients for early life.
- Iodine deficiency disorders once affected hundreds of millions, especially in regions with iodine-poor soil and water.
- Universal salt iodization remains the most effective public-health strategy, preventing goiter, hypothyroidism, and cretinism worldwide.
- Excess iodine can also disrupt thyroid function, triggering hyperthyroidism in susceptible individuals.
- Elemental iodine is toxic in gram quantities, while iodide is generally safe within recommended limits.
- Iodine’s medical role spans two centuries, from early tinctures to modern imaging agents and targeted radionuclide therapies.
- Radioactive isotopes such as I-123, I-125, and I-131 remain indispensable tools for diagnosing and treating thyroid disease and certain cancers.
- Lugol’s iodine continues to aid cervical cancer screening, revealing abnormal tissue by its failure to stain.
- Iodine chemistry still surprises, from its blue reaction with starch to its ability to form both I⁻ and I⁺ ions within biological systems.
- Despite global progress, iodine deficiency persists, especially in regions where iodized salt use is inconsistent or poorly regulated.
- Ensuring adequate iodine intake remains a simple, powerful intervention for protecting cognitive development and lifelong metabolic health.
(Cf. previous blogs entitled as: “Selenium: The Moon-Named Element That Protects Human Life.”; “Zinc: The Essential Element We Cannot Live Without.”)
Summary and Conclusions
Iodine remains one of the most paradoxical elements in human biology—needed in only microgram quantities, yet capable of influencing the health, cognition, and developmental trajectory of entire populations. Its essential role in the synthesis of T₄ and T₃ places it at the center of fetal brain development, metabolic regulation, and lifelong endocrine stability. When iodine is lacking, the consequences are profound: iodine deficiency continues to be the leading preventable cause of intellectual disability worldwide, a reminder of how fragile human development can be in the absence of this trace mineral.
The global introduction of iodized salt stands as one of the most successful public-health interventions of the 20th century, dramatically reducing goiter, hypothyroidism, and cretinism in many regions. Yet this success is uneven. Deficiency persists where iodization programs are poorly implemented, where soil and water remain iodine-poor, or where dietary patterns shift away from iodine-rich foods. These gaps highlight the ongoing need for consistent monitoring, policy enforcement, and public-health vigilance to ensure that iodine sufficiency is maintained across generations.
Despite centuries of use, important scientific questions remain unanswered. The precise molecular mechanisms governing iodine transport into the thyroid, the potential role of I⁺ intermediates in hormone synthesis, and the long-term effects of chronic low-grade deficiency are still being explored. Emerging dietary trends—particularly plant-based diets—may inadvertently reduce iodine intake unless carefully supplemented, raising new concerns for populations that already hover near the threshold of deficiency. These knowledge gaps point toward future research priorities, including the development of more sensitive biomarkers beyond urinary iodine concentration and a deeper understanding of how environmental contaminants such as nitrates, perchlorates, and thiocyanates interfere with iodine uptake.
Iodine’s medical relevance continues to expand. Its radioisotopes—I-123, I-125, and I-131—remain indispensable tools in imaging, cancer therapy, and thyroid disease management. Its chemistry still surprises, from its vivid blue reaction with starch to its ability to exist in multiple oxidation states that may influence hormone biosynthesis. These properties underscore iodine’s unique position at the intersection of chemistry, medicine, and public health.
Ultimately, iodine is far more than a nutrient. It is a quiet architect of human development, a stabilizing force in endocrine physiology, and a reminder that even the smallest elements can exert extraordinary influence on human life. Ensuring adequate iodine intake—through diet, supplementation, and sustained public-health efforts—remains one of the simplest and most powerful strategies for protecting cognitive potential and metabolic well-being across the lifespan.
For information on autism monitoring, screening and testing please read our blog.
References
- Ristic-Medic D, Piskackova Z, Hooper L, Ruprich J, Casgrain A, Ashton K, Pavlovic M, Glibetic M. Methods of assessment of iodine status in humans: a systematic review. Am J Clin Nutr. 2009 Jun;89(6):2052S-2069S. doi: 10.3945/ajcn.2009.27230H. Epub 2009 May 6. PMID: 19420096.
hts://pubmed.ncbi.nlm.nih.gov/19420096/tp
(A foundational review of biomarkers used to assess iodine status across populations.) - Hatch-McChesney A, Lieberman HR. Iodine and Iodine Deficiency: A Comprehensive Review of a Re-Emerging Issue. Nutrients. 2022 Aug 24;14(17):3474. doi: 10.3390/nu14173474. PMID: 36079737; PMCID: PMC9459956.
https://pubmed.ncbi.nlm.nih.gov/36079737/
(A modern, comprehensive analysis of global iodine deficiency trends, including developed nations.) - Meletis, Chris. (2011). Iodine Health Implications of Deficiency. Journal of Evidence-Based Complementary & Alternative Medicine. 16. 190-194. 10.1177/2156587211414424.
https://journals.sagepub.com/doi/epub/10.1177/2156587211414424
(Discusses the physiological consequences of deficiency and environmental antagonists such as perchlorate and bromine.)</strong> - World Health Organization (WHO). Assessment of Iodine Deficiency Disorders and Monitoring Their Elimination: A Guide for Programme Managers. 3rd ed. WHO Press.
https://www.who.int/publications/i/item/9789241595827
https://iris.who.int/server/api/core/bitstreams/82b512a6-57b8-45f3-8539-30b2f7f63564/content
(The global gold-standard reference for iodine deficiency surveillance and iodization programs.) - Zimmermann MB. Iodine deficiency. Endocr Rev. 2009 Jun;30(4):376-408. doi: 10.1210/er.2009-0011. Epub 2009 May 21. PMID: 19460960.
https://pubmed.ncbi.nlm.nih.gov/19460960/
(A widely cited review covering iodine physiology, deficiency disorders, and global epidemiology.) - Leung AM, Pearce EN, Braverman LE. Iodine nutrition in pregnancy and lactation. Endocrinol Metab Clin North Am. 2011 Dec;40(4):765-77. doi: 10.1016/j.ecl.2011.08.001. PMID: 22108279; PMCID: PMC3266621.
https://pubmed.ncbi.nlm.nih.gov/22108279/
(Essential for understanding maternal–fetal iodine requirements and neurodevelopmental outcomes.)