
Download Download & share this Knowledge card in your network [Free Download]
Table of Contents
- Introduction
- Understanding Depression: A Complex Condition
- Biological Basis of Depression
- Key Roles of Folate in Brain Function
- Folate and Depression: Research Insights
- Folate Transport in the Brain
- Folate Receptor Autoantibodies and Their Role
- Potential Treatment Approach: Folinic Acid (Leucovorin)
- Conclusion
- Did You Know About Folate Receptor Autoantibodies (FRAAs) and Brain Development?
Introduction
Depression is a major cause of morbidity and is the second-leading cause of disability world-wide. It is estimated that its prevalence is up to 15% of the population in industrialized nations and affects approximately 350 million people. By 2030, the World Health Organization projects it to be the leading cause of disease burden globally. Major Depressive Disorder is characterized by a state of low mood and anhedonia, affecting a person’s thoughts, behavior, feelings, and sense of well-being. Depressed patients may also present with diverse symptoms including lethargy, insomnia, social withdrawal and sexual dysfunction among a range of other ailments.
Understanding Depression: A Complex Condition
Depression is a complex and multifaceted mental health condition. It involves a combination of biological, psychological, and environmental factors, making it challenging to understand and treat. However, continued progress has been made and continues to be made in an attempt to elucidate its complex nature.
Biological Basis of Depression
Clearly, depression has several biological underpinnings. We know that changes in neurotransmitter levels, brain structure and function, genetics, and hormonal imbalances can contribute to the development of depression. For example, imbalances in neurotransmitters like serotonin and dopamine are often associated with depressive symptoms. We also know that Vitamin B-9, also known as folate, is essential for several bodily functions, including its important role in mental health and depression. There is a distinct connection between folate (Vitamin b-9) and multiple functions in the brain.
Key Roles of Folate in Brain Function
Distinctly, folate plays major roles in:
- Neurotransmitter Regulation: Folate plays a crucial role in the synthesis and regulation of neurotransmitters in the brain, such as serotonin, dopamine, and norepinephrine. These neurotransmitters are involved in mood regulation, and imbalances in them are linked to depression. Adequate folate levels can help ensure the proper functioning of these neurotransmitter systems.
- Methylation Process: Folate is involved in a biochemical process called methylation, which is essential for various cellular functions, including the regulation of gene expression. Methylation is crucial for maintaining normal brain function and may be disrupted in individuals with depression. Folate is required to produce S-adenosylmethionine (SAMe), a molecule involved in methylation reactions.
- Homocysteine Regulation: Folate, along with vitamins B-6 and B-12, helps to convert homocysteine, an amino acid, into methionine. Elevated homocysteine levels have been associated with an increased risk of depression and other mood disorders. Adequate folate intake can help keep homocysteine levels in check.
- Antioxidant Activity: Folate also has antioxidant properties, which means it can help protect cells, including brain cells, from oxidative stress and damage. Oxidative stress is thought to play a role in the development and progression of depression.
- Treatment Augmentation: In some cases, individuals with depression may benefit from folate supplementation in addition to their standard antidepressant treatment. Research has shown that adding folate to the treatment regimen of individuals who are not responding well to antidepressants may improve their response to therapy.
Download Download & share this infograph in your network [Free Download]
Folate and Depression: Research Insights
The relationship between folate and depression is complex, and multiple factors can contribute to the development of depression. It has been made clear, however, that folate does play a major role in this condition.
Several studies dating back to the 1960s have shown that depressed patients have a higher incidence of folate deficiency. In many studies investigating patients with neuropsychiatric disorders, folate deficiency was associated with low levels of the serotonin metabolite 5-hydroxyindoleacetic acid (5-HIAA) in the cerebrospinal fluid (CSF). Supplementation with folate seemed to restore CSF 5-HIAA levels to normal. Interestingly, there also were decreases in serotonin synthesis in patients with 5,10-methylenetetrahydrofolate reductase (MTHFR) deficiency (a genetic polymorphism disorder of folate metabolism). While the mechanisms regulating folate deficiency are not well known, low serotonin is implicated. This may involve S-adenosylmethionine (SAMe). SAMe is a major methyl donor formed from methionine. Folate is involved in a cycle that regenerates methionine from homocysteine after SAMe is demethylated to S-adenosylhomocysteine, with subsequent conversion to homocysteine. Folate deficiency decreases SAMe in brain.
Folate Transport in the Brain
With this in mind, we understand that importance of folate and its interconnections with neurotransmitter function. Folate, however, needs to be transported seamlessly into the cells. This is primarily achieved through several major folate receptors known as Folate Receptor Alpha (Fra), Reduced Folate Carrier (RFC), and the Proton Coupled Folate Transporter (PCFT). Folate Receptor Alpha (Fra) is a major receptor in transporting folate into the brain and cerebrospinal fluid (CSF). FRα acts as a high-affinity receptor for folate, facilitating its uptake into these cells. Cerebral folate delivery primarily occurs at the choroid plexus through FRα and the PCFT. If these transport systems are impeded in any way, low folate levels in the cerebrospinal fluid may occur thereby potentially causing neuropsychiatric disorders.
Folate Receptor Autoantibodies and Their Role
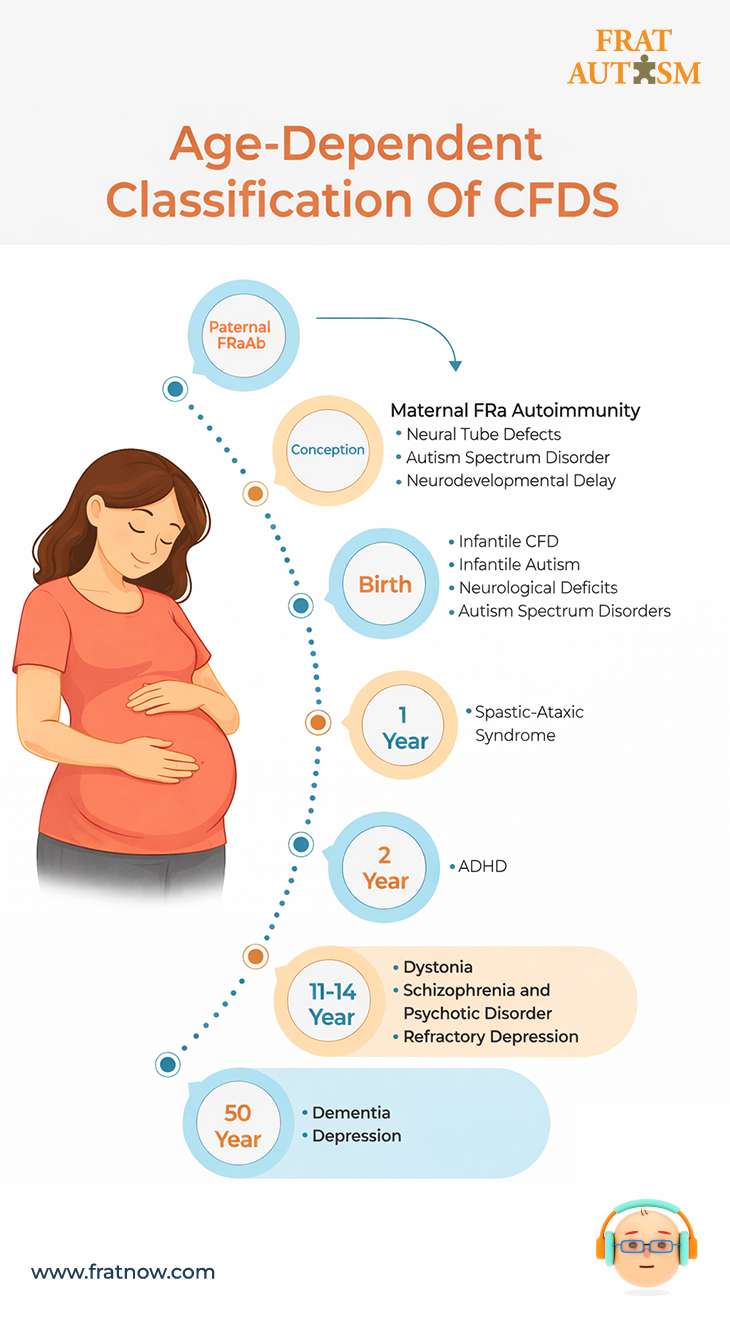
One impediment in the transport of folate to the brain and the CFS has been found to be the presence of folate receptor autoantibodies that target the Folate Receptor Alpha (Fra). Folate receptor autoantibodies make the Folate Receptor Alpha dysfunctional and suggest that not enough folate is being transported into the brain and into the cerebrospinal fluid. Researchers have defined this condition as cerebral folate deficiency syndrome. Interestingly, folate receptor autoantibodies have been found in patients with treatment resistant depression. In a previous study of 16 adult patients diagnosed with major depressive disorder, 9 patients tested positive for folate receptor autoantibodies. Although the study was small in the number of patients enrolled, it has garnered significant interest in assessing the relevance of these autoantibodies in depression. Screening for folate receptor autoantibodies may be done though the use of FRAT® (Folate Receptor Autoantibody Test).
Potential Treatment Approach: Folinic Acid (Leucovorin)
The presence of folate receptor autoantibodies in these instances also suggest a possible treatment that can possibly correct this type of impeded folate transport. More specifically, a special type of reduced folate known as folinic acid (leucovorin) has been found to reverse certain neuropsychiatric abnormalities in those that had folate receptor autoantibodies. It is believed that a reduced folate such as folinic acid can bypass the dysfunctional Folate Receptor Alpha and enter the brain/CFS via the Reduced Folate Carrier (RFC). However, RFC has a lower affinity for folate as compared with the FRα, so blood folate concentrations need to be higher to optimize this alternative pathway. This approach can be a possible solution for these depressed patients and may potentially serve as a future treatment. More research needs to be done in the fascinating field!
For information on autism monitoring, screening and testing please read our blog.




