Table of Contents
- Introduction
- Selenium — The Moon’s Element That Guards Human Life
- Selenium: The Moon-Named Element That Shapes Life
- The Fine Line Between Necessity and Toxicity
- Selenium in Food and Human Health
- Where Deficiency, Disease, and Human Physiology Intersect
- The Unexpected Defender Against Heavy Metals
- Take-Home Messages
- Summary and Conclusions
- Did You Know About Folate Receptor Autoantibodies (FRAAs) and Brain Development?
- References
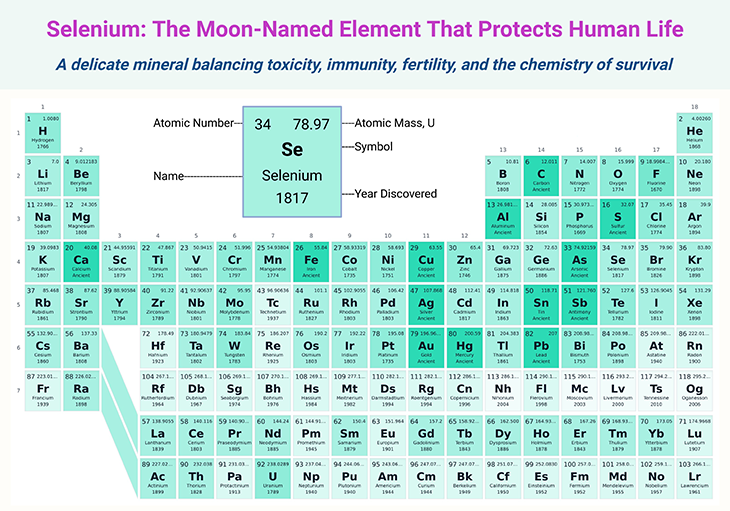
Figure 1. Zinc: The Essential Element We Cannot Live Without. From cosmic origins to human health – why this trace metal shapes growth, immunity, and lifelong development. [Adapted and modified from: https://pubchem.ncbi.nlm.nih.gov/periodic-table/]
Introduction
Selenium — The Moon’s Element That Guards Human Life
Selenium carries a name born from the heavens—drawn from Selene, the Greek goddess of the Moon—yet its influence is felt in every corner of human biology. It is an element of paradoxes: essential yet potentially toxic, rare in the Earth’s crust yet present in every cell of the human body, subtle in its daily actions yet capable of shaping health, development, and survival in profound ways.
Its scientific story began to unfold only in the late 20th century. In 1975, researchers discovered that selenium is a critical component of glutathione peroxidase, the enzyme that shields cells from destructive peroxides before they can ignite cascades of free-radical damage. A decade later, selenium emerged again at the center of human physiology when it was found in deiodinase, the enzyme that activates thyroid hormones and governs metabolic rhythm. These discoveries transformed selenium from a chemical curiosity into a cornerstone of antioxidant defense, endocrine balance, and cellular protection.
But selenium’s reach extends even further. Nature has woven it into at least 14 selenoproteins, many of which are indispensable for sperm formation, embryonic development, and immune resilience. Its presence in foods—whether as selenocysteine in garlic and broccoli or selenomethionine in grains and meat—reflects the delicate interplay between soil, agriculture, and human nutrition. Too little selenium invites vulnerability: weakened immunity, heart dysfunction, impaired fertility, and region-specific diseases such as Keshan disease. Too much, however, tips the balance toward toxicity, with unmistakable signs like foul breath, dermatitis, and systemic poisoning [1-6].
And then comes selenium’s most surprising role: its ability to neutralize some of the world’s most dangerous heavy metals. Mercury, cadmium, arsenic, and thallium—elements that devastate enzymes and tissues—are disarmed by selenium’s stronger chemical pull. Even mercury-rich tuna remains safe to eat because the fish binds one selenium atom for every mercury atom, forming protective complexes that prevent biochemical harm.
Selenium is, in every sense, an element of contrasts—a guardian and a threat, a nutrient and a toxin, a trace mineral with outsized influence. Its story reminds us that life depends not only on abundance, but on balance; not only on the elements we see, but on those that work quietly in the background, shaping health in ways both subtle and extraordinary.
Selenium: The Moon-Named Element That Shapes Life
Origins and Etymology
Pronounced “sel-ee-nee-um,” the element draws its name from Selene, the Greek goddess of the Moon. The lunar reference is fitting: selenium is both mysterious and essential, quietly influencing life, technology, and planetary chemistry.
Life’s Quiet Guardian in Every Cell
Selenium’s importance spans an astonishing range—from sperm biology to semiconductor technology, from soil fertility to the manufacture of specialized glass. Its versatility reflects a unique chemical profile that allows selenium to participate in both biological and industrial systems with remarkable efficiency.
Element of Life
Selenium’s biological significance was firmly established in 1975, when Yogesh Awasthi at Galveston, Texas, demonstrated that selenium is an integral component of glutathione peroxidase, a major antioxidant enzyme that neutralizes peroxides before they generate harmful free radicals. This discovery placed selenium at the center of the body’s oxidative defense network.
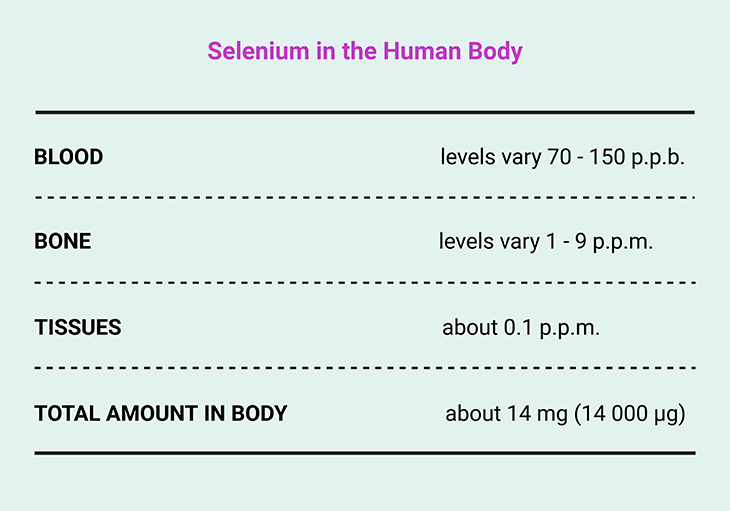
Table 1. Element of Life ~ Selenium in the Human Body.
Further insight came in 1991, when Professor Dietrich Behne at the Hahn-Meitner Institute in Berlin identified selenium within deiodinase, the enzyme responsible for activating thyroid hormones. This finding linked selenium directly to thyroid function, metabolism, and energy regulation.
Today, it is known that every human cell contains more than one million atoms of selenium. The highest concentrations occur in hair, kidneys, and testicles, reflecting selenium’s roles in detoxification, filtration, and reproduction. Yet selenium presents a familiar nutritional paradox: too little impairs health, but too much becomes toxic.
Nature synthesizes selenium in the form of methylselenocysteine, a selenium-containing amino acid found in Brassica vegetables, garlic, and other Allium species. Remarkably, the genetic code includes a specialized segment of messenger RNA that directs the incorporation of selenium into proteins. To date, approximately 14 selenoproteins have been identified. While they function in various tissues—including the kidneys—their most critical roles lie in sperm development and the early embryo, underscoring selenium’s importance in fertility and early life [1-6] (see Table 1).
The Fine Line Between Necessity and Toxicity
Despite its essentiality, selenium can be profoundly hazardous in certain forms. Hydrogen selenide (H₂Se) is extremely toxic and nearly killed the chemist who first synthesized it.
Even dietary selenium can become dangerous when consumed in excess. A dose of 5 mg is highly toxic, and chronic low-level exposure—historically, seen in certain industrial settings—caused weight loss, anemia, dermatitis, and severe social isolation due to the unbearable odor emitted by affected individuals. The recommended maximum daily intake is 0.45 mg (450 μg); exceeding this threshold risks selenium poisoning, most notably characterized by extremely foul breath and body odor. These odors arise from volatile methylated selenium compounds produced as the body attempts to eliminate the excess.
A dramatic example of selenium toxicity occurred in April 2009 at the prestigious U.S. polo championships. Horses were given a mineral supplement called Biodyl, intended to provide selenium, magnesium, potassium, and B vitamins. Due to a compounding error, the batch contained excessive selenium, and within three hours of ingestion, the horses began to collapse. All 21 horses that received the supplement died from acute selenium poisoning—a stark reminder of the razor-thin line between selenium’s essentiality and its lethality.
Selenium in Food and Human Health
From Earth to Plate: How Diet Shapes Selenium Status
In the human diet, selenium appears primarily in two organic forms: selenocysteine, found in foods such as broccoli, garlic, and other Allium and Brassica vegetables, and selenomethionine, which predominates in meat and grain products. Structurally, selenocysteine contains one fewer carbon atom than selenomethionine, and unlike selenomethionine, it is not incorporated into proteins in place of methionine. This biochemical distinction influences how selenium is stored, metabolized, and mobilized within the body.
Daily selenium intake varies widely—from 6 to 200 μg, depending on dietary patterns. The average Westerner consumes about 65 μg per day, an amount sufficient to prevent deficiency. This intake falls slightly below the recommended 75 μg/day for men, but meets the recommended 60 μg/day for women. Although selenium losses may occasionally exceed dietary absorption, this does not pose immediate risk because the body can draw upon selenium reserves stored in bone.
As a supplement, selenium is commonly taken as sodium selenite (Na₂SeO₃), a water-soluble white crystalline compound, typically at a dose of 50 μg per day. Another natural option is brewer’s yeast grown on selenium-enriched media, which accumulates selenium in organic forms.
For most individuals, selenium intake comes from breakfast cereals and bread, particularly whole-meal bread, where two slices can provide 30 μg. However, selenium content in wheat and meat varies significantly depending on the selenium levels in the soil where crops or livestock were raised.
Some foods are exceptionally rich in selenium. Brazil nuts and molasses (black treacle) contain >100 μg per 100 g, while seafoods such as tuna, cod, and salmon, and organ meats like liver and kidney, exceed 30 μg per 100 g. Many nuts—including peanuts and cashews—contain similar amounts; a 100-gram bag of cashews may provide 65 μg. Additional selenium-dense foods include wheat germ, bran, and brewer’s yeast [1-6].
One of the richest natural sources is the edible mushroom Albatrellus pes-caprae, popular in Italy, which contains an extraordinary 3,700 μg per 100 g (fresh weight). A single dish of this size would deliver eight times the recommended daily maximum. Fortunately, much of the selenium in mushrooms is poorly absorbed, making them safe to consume despite their high content.
Where Deficiency, Disease, and Human Physiology Intersect
Selenium is indispensable for human health, and inadequate levels have been associated with cancer, heart disease, and infertility. Additional conditions linked to selenium deficiency include anemia, hypertension, arthritis, premature aging, muscular dystrophy, multiple sclerosis, and AIDS. Selenium strongly influences the activity of T-lymphocytes, the immune system’s natural killer cells, which defend the body and can even destroy tumor cells.
The relationship between selenium and cancer prevention has been extensively studied. Epidemiological data suggest a protective effect, though findings are not universally consistent. In one U.S. trial involving 1,300 elderly participants over four years, selenium supplementation was associated with a 30% reduction in new cases of lung, bowel, and prostate cancer. While such results are compelling, other studies have not replicated these benefits, highlighting the complexity of selenium’s role in carcinogenesis.
In certain diseases, however, the evidence for selenium deficiency is unequivocal. In regions of China such as Keshan and Linxian, where soils are severely depleted of selenium, children in the Keshan region suffered from a form of congestive heart failure known as Keshan disease. Residents of Linxian experienced unusually high rates of stomach cancer. In both cases, the incidence of disease declined markedly when selenium supplements were provided to the population [1-6].
Selenium also plays a critical role in male fertility. Some researchers attribute the declining sperm counts observed among European men to reduced dietary selenium intake. Over the past century, average selenium consumption in Europe has fallen by half, partly due to decreased consumption of selenium-rich foods such as kidney and liver, and partly because European wheat is grown in selenium-poor soils. A double-blind trial provided strong evidence for selenium’s reproductive importance: men receiving selenium supplements doubled their output of viable sperm, while those given a placebo showed no improvement.
The Unexpected Defender Against Heavy Metals
Selenium is full of paradoxes. Although the element itself can be toxic even at relatively low doses, it possesses the remarkable ability to counteract the toxicity of other heavy metals—most notably cadmium, mercury, arsenic, and thallium. Because of this, selenium is often described as a biological antagonist to these metals, capable of neutralizing or diminishing their harmful effects.
One of the most striking examples comes from tuna, a species known to accumulate higher-than-expected levels of mercury. Despite this, tuna remains generally safe to eat. The prevailing explanation is that the fish protects itself by absorbing one atom of selenium for every atom of mercury it takes in. This molecular pairing prevents mercury from exerting its full toxic potential.
The protective mechanism is elegantly chemical. Mercury binds strongly to selenium—even more strongly than it binds to sulfur, the element it typically attaches to when poisoning enzymes. In mercury toxicity, the metal disrupts enzyme function by binding to sulfur-containing amino acids at critical catalytic sites. Selenium intervenes by pulling mercury away from these enzyme sites, forming stable mercury–selenium complexes. Once mercury is removed, the previously blocked enzymes can resume normal function, restoring essential biochemical pathways.
This unexpected defensive role—where a potentially toxic element becomes a guardian against even more dangerous toxins—captures the essence of selenium’s dual nature. It is an element capable of harm, yet indispensable for protection; a micronutrient that, in the right balance, shields life from some of the most potent poisons in the natural world.
Take-Home Messages
- Selenium is essential for human biology, yet harmful when deficient or excessive.
- Every cell contains over a million selenium atoms, highlighting its deep biological integration.
- Selenium supports antioxidant defense through glutathione peroxidase and protects thyroid function via deiodinase.
- Fertility depends on selenium, especially for healthy sperm and early embryonic development.
- Daily intake varies widely, from 6 to 200 μg depending on diet.
- Most Western diets provide about 65 μg/day, enough to prevent deficiency for most adults.
- Soil selenium levels strongly influence food content, especially in grains and meat.
- Foods like Brazil nuts, seafood, organ meats, and whole grains are reliable selenium sources.
- Selenium deficiency contributes to serious diseases, including Keshan disease, heart problems, and impaired immunity.
- Some studies show a 30% reduction in certain cancers with supplementation, though results remain mixed.
- Low selenium intake may reduce sperm count, and supplementation can improve viable sperm output.
- Selenium toxicity occurs above 0.45 mg/day, with foul breath and body odor as hallmark signs.
- Hydrogen selenide is extremely toxic, underscoring selenium’s dual nature.
- Acute poisoning can be fatal, as seen in the 2009 polo horse tragedy.
- Selenium protects against heavy metals, especially mercury, cadmium, arsenic, and thallium.
- Mercury binds more strongly to selenium than to sulfur, allowing selenium to free blocked enzymes.
- Tuna remains safe to eat partly because it balances mercury with selenium atom-for-atom.
- Selenium embodies biological balance—too little harms, too much harms, but the right amount sustains life in remarkable ways.
(Cf. previous blog entitled as: “Zinc: The Essential Element We Cannot Live Without.”)
Summary and Conclusions
Selenium stands out as a paradoxical yet indispensable trace element—essential for antioxidant defense, thyroid hormone activation, immune function, fertility, and early embryonic development, yet capable of toxicity when intake exceeds a narrow safe range. Its biological influence is anchored in at least 14 selenoproteins, each contributing to cellular protection, metabolic balance, or reproductive success. The presence of over a million selenium atoms in every human cell underscores its deep evolutionary integration into human physiology.
Dietary selenium intake varies widely across the globe, shaped by soil composition, agricultural practices, and changing dietary patterns. This variability leaves certain populations vulnerable to deficiency-related disorders such as Keshan disease, impaired immunity, reduced sperm quality, and possibly increased risks of specific cancers. At the same time, selenium’s ability to neutralize heavy metals—including mercury, cadmium, arsenic, and thallium—reveals a protective role that extends beyond nutrition into toxicology and environmental health.
Despite decades of research, important gaps remain. The full catalog of human selenoproteins is still incomplete, and current biomarkers of selenium status lack precision, making it difficult to distinguish between adequacy, marginal deficiency, and early toxicity. The relationship between selenium and cancer prevention remains inconsistent across studies, suggesting complex interactions with genetics, diet, and environmental exposures. Additionally, the long-term effects of mild chronic deficiency or low-grade excess are not well understood, particularly in vulnerable groups such as pregnant women, infants, and individuals with chronic illness.
Future research must focus on developing more accurate biomarkers, clarifying selenium’s role in neurodevelopment and immune aging, and understanding how genetic variations in selenium metabolism influence individual requirements. There is also a growing need to refine personalized supplementation strategies, especially in regions with selenium-depleted soils or in populations with increased physiological demands. As we deepen our understanding, selenium continues to exemplify the delicate balance of essential elements—one that sustains human health through mechanisms both expected and profoundly surprising.
For information on autism monitoring, screening and testing please read our blog.
References
- Rayman MP. Selenium and human health. Lancet. 2012 Mar 31;379(9822):1256-68. doi: 10.1016/S0140-6736(11)61452-9. Epub 2012 Feb 29. PMID: 22381456.
https://pubmed.ncbi.nlm.nih.gov/22381456/
(A landmark review covering selenium biology, deficiency, toxicity, selenoproteins, and global health implications.) - Steinbrenner H, Speckmann B, Klotz LO. Selenoproteins: Antioxidant selenoenzymes and beyond. Arch Biochem Biophys. 2016 Apr 1;595:113-9. doi: 10.1016/j.abb.2015.06.024. PMID: 27095226.
https://pubmed.ncbi.nlm.nih.gov/27095226/
(Defines the roles of major selenoproteins, including glutathione peroxidases and thioredoxin reductases.) - Fairweather-Tait SJ, Bao Y, Broadley MR, Collings R, Ford D, Hesketh JE, Hurst R. Selenium in human health and disease. Antioxid Redox Signal. 2011 Apr 1;14(7):1337-83. doi: 10.1089/ars.2010.3275. Epub 2011 Jan 6. PMID: 20812787.
https://pubmed.ncbi.nlm.nih.gov/20812787/
(A comprehensive synthesis of selenium metabolism, biomarkers, dietary sources, and clinical outcomes.) - Vinceti M, Filippini T, Rothman KJ. Selenium exposure and the risk of type 2 diabetes: a systematic review and meta-analysis. Eur J Epidemiol. 2018 Sep;33(9):789-810. doi: 10.1007/s10654-018-0422-8. Epub 2018 Jul 5. PMID: 29974401.
https://pubmed.ncbi.nlm.nih.gov/29974401/
(Explores the nuanced relationship between selenium status and metabolic disease risk.) - Navarro-Alarcon M, Cabrera-Vique C. Selenium in food and the human body: a review. Sci Total Environ. 2008 Aug 1;400(1-3):115-41. doi: 10.1016/j.scitotenv.2008.06.024. Epub 2008 Jul 26. PMID: 18657851.
https://pubmed.ncbi.nlm.nih.gov/18657851/
(Focuses on selenium content in foods, bioavailability, and global variability due to soil differences.) - Hatfield DL, Tsuji PA, Carlson BA, Gladyshev VN. Selenium and selenocysteine: roles in cancer, health, and development. Trends Biochem Sci. 2014 Mar;39(3):112-20. doi: 10.1016/j.tibs.2013.12.007. Epub 2014 Jan 28. PMID: 24485058; PMCID: PMC3943681.
https://pubmed.ncbi.nlm.nih.gov/24485058/
(A mechanistic deep dive into selenocysteine biology, cancer pathways, and developmental roles.)